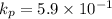Chemistry, 28.07.2021 04:30 markcardenas2756
Hydrogen chloride decomposes to form hydrogen and chlorine, like this:
2HCl(g) + H2(g) â Cl2(g)
Also, a chemist finds that at a certain temperature the equilibrium mixture of hydrogen chloride, hydrogen, and chlorine has the following composition:
compound pressure at equilibrium
HCl 84.4 atm
H2 77.9 atm
Cl2 54.4
Required:
Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1


Chemistry, 22.06.2019 21:00
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1

Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3
You know the right answer?
Hydrogen chloride decomposes to form hydrogen and chlorine, like this:
2HCl(g) + H2(g) â Cl2(g)
Questions






Chemistry, 02.07.2019 19:10














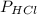 = 84.4 atm
= 84.4 atm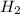 ,
, 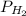 = 77.9 atm
= 77.9 atm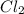 ,
, 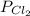 = 54.4 atm
= 54.4 atm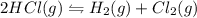
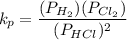
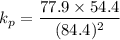
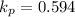
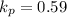 (in 2 significant figures)
(in 2 significant figures)