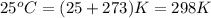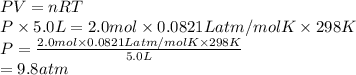Chemistry, 13.05.2021 09:40 laylac45531
A sample of 2.0 moles of helium gas is contained in a tank with a volume of 5.0L at a temperature of 25°C. What is the pressure of the gas in the tank
in atm?
Given: R = 0.0821 L. atm/mol. K
O 9.8 atm
O 0.00069 atm
O 0.82 atm
O 0.0082 atm

Answers: 3


Another question on Chemistry




You know the right answer?
A sample of 2.0 moles of helium gas is contained in a tank with a volume of 5.0L at a temperature of...
Questions


Computers and Technology, 10.10.2019 05:10