+
For the reaction Pb(NO3)2 + 2KI Pbl2 + 2KNO3, how many moles of lead iodide are
produced fr...


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1
You know the right answer?
Questions

Chemistry, 19.10.2019 22:30

Mathematics, 19.10.2019 22:30


Mathematics, 19.10.2019 22:30

English, 19.10.2019 22:30


Biology, 19.10.2019 22:30

Advanced Placement (AP), 19.10.2019 22:30

Social Studies, 19.10.2019 22:30

Biology, 19.10.2019 22:30


History, 19.10.2019 22:30


History, 19.10.2019 22:30

Mathematics, 19.10.2019 22:30

Social Studies, 19.10.2019 22:30


Mathematics, 19.10.2019 22:30

Social Studies, 19.10.2019 22:30

Advanced Placement (AP), 19.10.2019 22:30

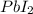 will be produced from 237.1 g of potassium iodide
will be produced from 237.1 g of potassium iodide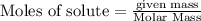
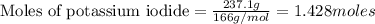
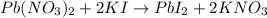
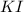 produce = 1 mole of
produce = 1 mole of 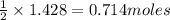 of
of 

