
Chemistry, 31.03.2020 01:29 jaqwannewsome
A metal object with mass of 23.2 g 23.2 g is heated to 97.0 °C 97.0 °C and then transferred to an insulated container containing 90.0 g 90.0 g of water at 20.5 °C. 20.5 °C. The water temperature rises and the temperature of the metal object falls until they both reach the same final temperature of 22.6 °C. 22.6 °C. What is the specific heat of this metal object? Assume that all the heat lost by the metal object is absorbed by the water.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1

Chemistry, 23.06.2019 09:30
What is the best describtion of the side of the moon that faces earth?
Answers: 2

Chemistry, 23.06.2019 11:00
Which of the following reactions is endothermic? h2(g) + ½ o2(g) h2o(g), h = -57.82 kcal ½n2(g) + o2(g) + 8.1 kcal no2(g) ½ n2(g) + 3/2 h2(g) nh3(g) + 11.0 kcal c(diamond) + o2(g) co2, h = -94.50 kcal
Answers: 2

Chemistry, 23.06.2019 12:10
What is the correct name for hg(no3)2? mercury (i) nitrate mercury (ii) nitrate mercury nitroxide mercury dinitride
Answers: 1
You know the right answer?
A metal object with mass of 23.2 g 23.2 g is heated to 97.0 °C 97.0 °C and then transferred to an in...
Questions


Engineering, 27.09.2020 09:01



Mathematics, 27.09.2020 09:01

Mathematics, 27.09.2020 09:01


Mathematics, 27.09.2020 09:01


History, 27.09.2020 09:01

Geography, 27.09.2020 09:01

Social Studies, 27.09.2020 09:01



Chemistry, 27.09.2020 09:01

History, 27.09.2020 09:01

Mathematics, 27.09.2020 09:01


Mathematics, 27.09.2020 09:01

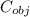 = 0.457
= 0.457 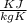
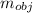 = 23.2 gm
= 23.2 gm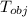 = 97 ° c
= 97 ° c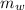 = 90 gm
= 90 gm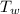 = 20.5 ° c
= 20.5 ° c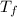 = 22.6 ° c
= 22.6 ° c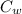 (
( 

