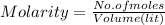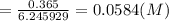Chemistry, 31.03.2020 01:28 rainbow7020
A sample of 8.4 grams of NaOH is dissolved into 620 mL of aqueous 0.250 M NaOH (assume no volume change). This solution is then poured into 1.65 gallons of water. (You may assume that the two volumes can be added.) What is the concentration of NaOH in the final solution

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1


Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
A sample of 8.4 grams of NaOH is dissolved into 620 mL of aqueous 0.250 M NaOH (assume no volume cha...
Questions








Mathematics, 10.12.2020 18:50

SAT, 10.12.2020 18:50


History, 10.12.2020 18:50



Mathematics, 10.12.2020 18:50

History, 10.12.2020 18:50





Mathematics, 10.12.2020 18:50

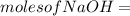
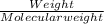
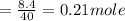 620 ml of aqueous 0.25 M NaOH
620 ml of aqueous 0.25 M NaOH 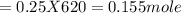 Total moles = 0.21 + 0.155 = 0.365 moles1.65 gallons = 3.785 lit x 1.65 = 6.245929 lit (∵ 1 gallon = 3.785 lit)
Total moles = 0.21 + 0.155 = 0.365 moles1.65 gallons = 3.785 lit x 1.65 = 6.245929 lit (∵ 1 gallon = 3.785 lit)