

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2



Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
Calculate the molarity of a kcl solution made by dissolving 21.2 g of kcl in a total volume of 500....
Questions

Business, 07.04.2022 02:00

Mathematics, 07.04.2022 02:10






Mathematics, 07.04.2022 02:40




Mathematics, 07.04.2022 03:10




Mathematics, 07.04.2022 03:30


Social Studies, 07.04.2022 03:40

Mathematics, 07.04.2022 03:50

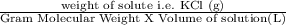
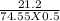 = 0.569 M
= 0.569 M

