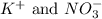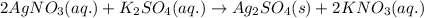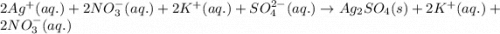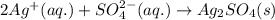Chemistry, 25.07.2019 03:30 Larkinlover703
What are the spectator ions in the solution after the precipitation of an insoluble sulfate compound from agno3(aq) and k2so4(aq)?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
What are the spectator ions in the solution after the precipitation of an insoluble sulfate compound...
Questions

Mathematics, 14.02.2022 15:40

Engineering, 14.02.2022 15:40






Computers and Technology, 14.02.2022 15:40

Mathematics, 14.02.2022 15:40






Business, 14.02.2022 15:50


Mathematics, 14.02.2022 15:50

Chemistry, 14.02.2022 15:50

Mathematics, 14.02.2022 15:50

Mathematics, 14.02.2022 15:50