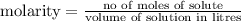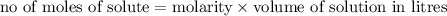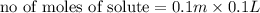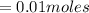Chemistry, 03.08.2019 01:30 alyssawage
Calculate the number of moles of kool aid powder needed to make 100ml of a 0.1 m solution? 0.1m=x mol c12h22o11/0.1l

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3


Chemistry, 23.06.2019 01:30
Concentrations expressed as a percent by mass are useful when the solute is a a. liquid b. gas c. solid
Answers: 1

Chemistry, 23.06.2019 07:10
Which one of the following is an oxidation-reduction reaction? naoh + hno3 --> h2o + kno3 naoh + hno3 --> h2o + kno3 so3 + h2o --> h2so4 cacl2 + na2co3 --> caco3 + 2 nacl ch4 + 2 o2 --> co2 + 2 h2o al2(so4)3 + 6 koh --> 2 al(oh)3 + 3 k2so4
Answers: 3
You know the right answer?
Calculate the number of moles of kool aid powder needed to make 100ml of a 0.1 m solution? 0.1m=x m...
Questions

History, 27.06.2020 18:01




Mathematics, 27.06.2020 18:01