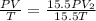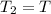Chemistry, 18.07.2019 17:40 ernestscott406
Consider a balloon that has a volume v. it contains n moles of gas, it has an internal pressure of p, and its temperature is t. if the balloon is heated to a temperature of 15.5t while it is placed under a high pressure of 15.5p, how does the volume of the balloon change?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 23.06.2019 06:20
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
Consider a balloon that has a volume v. it contains n moles of gas, it has an internal pressure of p...
Questions

Physics, 28.01.2020 01:31

Mathematics, 28.01.2020 01:31

History, 28.01.2020 01:31

English, 28.01.2020 01:31






Mathematics, 28.01.2020 01:31


Mathematics, 28.01.2020 01:31

English, 28.01.2020 01:31


Business, 28.01.2020 01:31




Mathematics, 28.01.2020 01:31

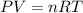
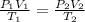
 = initial pressure
= initial pressure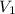 = initial volume
= initial volume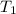 = initial temperature
= initial temperature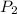 = final pressure
= final pressure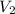 = final volume
= final volume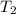 = final temperature
= final temperature