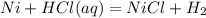A student dropped a piece of nickel metal into a solution of HCl(aq). He observed the formation of gas bubbles and collected the gas into another test tube. The student performed a splint test and observed that the splint flared up when he placed the splint into the test tube of the gas. What can be said about the results of this students experiment?
a. The student performed the splint test incorrectly. He should of observed a popping sound when the splint was placed in the test tube.
b. The experiment was performed incorrectly. Nickel doesn't react with HCl. Therefore, the student picked up the wrong metal when conducting the experiment.
c. The student completed the experiment correctly and there were no errors in the experiment.
d. The student performed the splint test incorrectly. He should of observed the flame being extinguished when the splint was placed in the test tube.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

You know the right answer?
A student dropped a piece of nickel metal into a solution of HCl(aq). He observed the formation of g...
Questions

Mathematics, 13.05.2021 05:10



Mathematics, 13.05.2021 05:10


Computers and Technology, 13.05.2021 05:10

Social Studies, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10

Chemistry, 13.05.2021 05:10

Biology, 13.05.2021 05:10

Mathematics, 13.05.2021 05:10


Mathematics, 13.05.2021 05:10

Computers and Technology, 13.05.2021 05:10


Mathematics, 13.05.2021 05:10