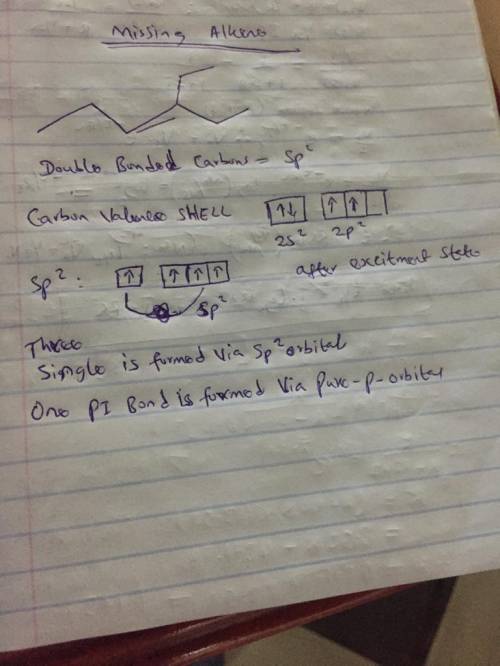Chemistry, 03.07.2021 04:10 hickslily9
We can use bond-line formulas to represent alkenes in much the same way that we use them to represent alkanes. Consider the following alkene: h5ch5e4 How many carbon atoms are sp2−hybridized in this alkene?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 23.06.2019 00:00
Predict the relative bond lengths of the three carbon-oxygen bonds in the carbonate ion (co2−3). what would you expect the charge to be on each oxygen? match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Answers: 3

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 2
You know the right answer?
We can use bond-line formulas to represent alkenes in much the same way that we use them to represen...
Questions


Mathematics, 10.03.2021 21:00

English, 10.03.2021 21:00


Mathematics, 10.03.2021 21:00


Mathematics, 10.03.2021 21:00

Chemistry, 10.03.2021 21:00






English, 10.03.2021 21:00


History, 10.03.2021 21:00

Mathematics, 10.03.2021 21:00

Physics, 10.03.2021 21:00

History, 10.03.2021 21:00