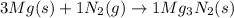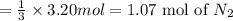Chemistry, 19.05.2021 16:00 aliami0306oyaj0n
When 3.20 moles of magnesium is heated and placed in a container of nitrogen gas the two substances undergo a synthesis reaction. How many moles of nitrogen gas will react? 3 Mg (s) + 1 N2 (g) → 1 Mg3N2 (s)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3

Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1

You know the right answer?
When 3.20 moles of magnesium is heated and placed in a container of nitrogen gas the two substances...
Questions








English, 29.12.2020 17:50





Biology, 29.12.2020 17:50



Mathematics, 29.12.2020 17:50

World Languages, 29.12.2020 17:50



Geography, 29.12.2020 18:00