
Chemistry, 15.05.2021 18:50 jasminebrown72
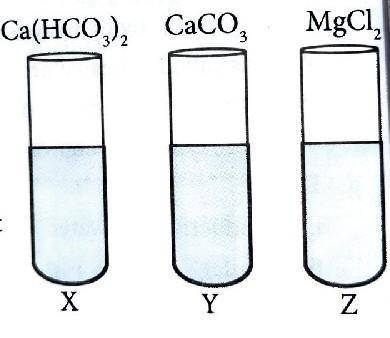
 X , Y and Z are the samples of water obtained from different sources. The types of salt dissolved in them are labelled in the picture. Now , Answer the following questions.
i. Identify the hardness of water that can be removed by boiling process.
X , Y and Z are the samples of water obtained from different sources. The types of salt dissolved in them are labelled in the picture. Now , Answer the following questions.
i. Identify the hardness of water that can be removed by boiling process.
ii. Which of them is soft water ?
iii. Write the chemical equation of the reaction that takes place when Z is treated with X , Y and Z.
~Random/Irrelevant answers will be reported!

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
X , Y and Z are the samples of water obtained from different sources. The types of salt dissolved i...
Questions


History, 16.07.2019 02:00





Mathematics, 16.07.2019 02:00


Health, 16.07.2019 02:00

Mathematics, 16.07.2019 02:00

Geography, 16.07.2019 02:00

History, 16.07.2019 02:00


Arts, 16.07.2019 02:00

Chemistry, 16.07.2019 02:00





Social Studies, 16.07.2019 02:00








