
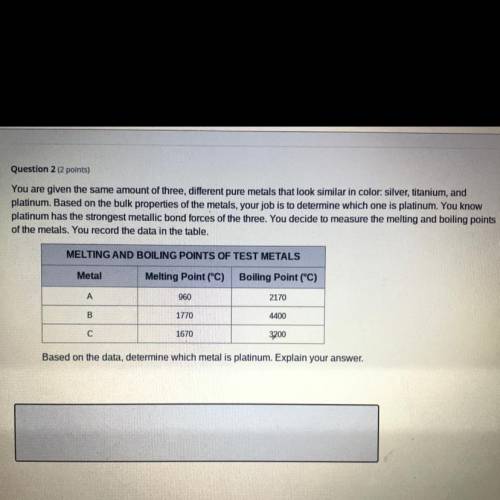
You are given the same amount of three different pure metals that look similar in color: silver, titanium, and platinum. Based on the bulk properties of the metals, your job is to determine which one is platinum. You know platinum has the strongest metallic bond forces of the three. You decide to measure the melting and boiling points of the metals. You record the data in the table.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

You know the right answer?
You are given the same amount of three different pure metals that look similar in color: silver, tit...
Questions

Mathematics, 24.09.2019 05:30

Biology, 24.09.2019 05:30




Biology, 24.09.2019 05:30

Mathematics, 24.09.2019 05:30


History, 24.09.2019 05:30


Mathematics, 24.09.2019 05:30


Mathematics, 24.09.2019 05:30





Health, 24.09.2019 05:30

Mathematics, 24.09.2019 05:30

Social Studies, 24.09.2019 05:30



