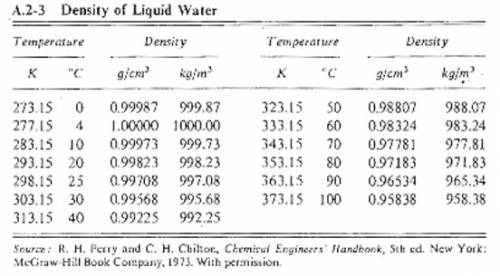
Suppose you were calibrating a 100.0 ml volumetric flask using distilled water. the flask temperature was at 20°c, and you assumed that the distilled water was as well. however, you later discover that the actual water temperature was 11°c instead. how is the mass of the 100.0 ml of distilled water you measured at 11°c different from the mass of 100.0 ml of distilled water at 20°c?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
Suppose you were calibrating a 100.0 ml volumetric flask using distilled water. the flask temperatur...
Questions


Mathematics, 05.04.2020 02:51



Biology, 05.04.2020 02:51

Mathematics, 05.04.2020 02:51

History, 05.04.2020 02:51




Mathematics, 05.04.2020 02:51



Social Studies, 05.04.2020 02:51

Mathematics, 05.04.2020 02:51

English, 05.04.2020 02:51



Mathematics, 05.04.2020 02:51