
Chemistry, 21.09.2019 22:00 SignoraPenguino
Which substance most likely has the highest boiling point?
calcium chloride (cacl2), an ionic compound
methanol (ch3oh), a polar covalent compound
carbon tetrachloride (ccl4), a nonpolar covalent compound

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3


Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1
You know the right answer?
Which substance most likely has the highest boiling point?
calcium chloride (cacl2), an ionic...
calcium chloride (cacl2), an ionic...
Questions


Advanced Placement (AP), 02.11.2020 19:30

English, 02.11.2020 19:30

Biology, 02.11.2020 19:30







English, 02.11.2020 19:30

Law, 02.11.2020 19:30

English, 02.11.2020 19:30

Computers and Technology, 02.11.2020 19:30




Mathematics, 02.11.2020 19:30


Mathematics, 02.11.2020 19:30

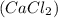 , an ionic compound.
, an ionic compound.

