
Chemistry, 11.04.2021 08:40 susannaking5852
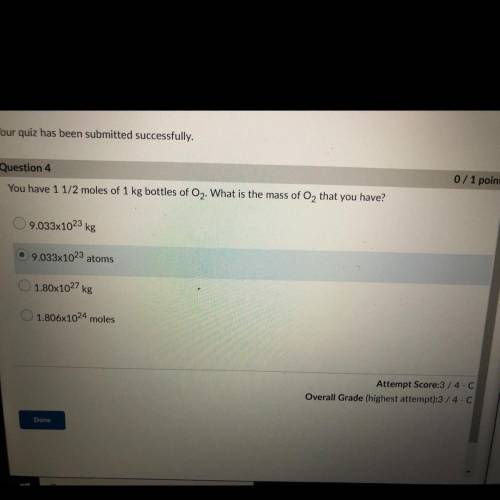
You have 1 1/2 moles of 1 kg bottles of O2. What is the mass of O2 that you have?
A. 9.033x10^23 kg
B. 9.033x10^23 atoms
C. 1.80x10^27 kg
D. 1.806x10^24 moles
It is not B as I have already tried it :(

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2


Chemistry, 23.06.2019 07:20
Which of the following are acids or bases? 1. sodium hydrogen 2. barium hydroxide solution 3. carbonate solution
Answers: 1

Chemistry, 23.06.2019 09:00
Chortling is used to clean water. another possible atom that would also work is a. sodium b. sulfur c. bromine
Answers: 1
You know the right answer?
You have 1 1/2 moles of 1 kg bottles of O2. What is the mass of O2 that you have?
A. 9.033x10^23 kg...
Questions


Advanced Placement (AP), 05.11.2019 16:31

Mathematics, 05.11.2019 16:31




History, 05.11.2019 16:31

Mathematics, 05.11.2019 16:31






Chemistry, 05.11.2019 16:31

Social Studies, 05.11.2019 16:31

Social Studies, 05.11.2019 16:31

Mathematics, 05.11.2019 16:31





