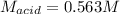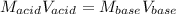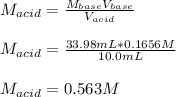Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodium hydroxide solution according to the equation H C 2 H 3 O 2 + N a O H ⟶ H 2 O + N a C 2 H 3 O 2 HCX2HX3OX2+NaOH⟶HX2O+NaCX2HX3OX2 If you require 33.98 mL of 0.1656 M N a O H NaOH solution to titrate 10.0 mL of H C 2 H 3 O 2 HCX2HX3OX2 solution, what is the molar concentration of acetic acid in the vinegar? Type

Answers: 2


Another question on Chemistry

Chemistry, 20.06.2019 18:04
If this equation was completed which statement would it best support
Answers: 1

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1
You know the right answer?
Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodi...
Questions

Social Studies, 17.07.2019 14:50

Social Studies, 17.07.2019 14:50


Social Studies, 17.07.2019 14:50

Mathematics, 17.07.2019 14:50



Mathematics, 17.07.2019 14:50

History, 17.07.2019 14:50

History, 17.07.2019 14:50

Mathematics, 17.07.2019 14:50

Social Studies, 17.07.2019 14:50

Biology, 17.07.2019 14:50

Mathematics, 17.07.2019 14:50

Social Studies, 17.07.2019 14:50

Biology, 17.07.2019 14:50

French, 17.07.2019 14:50


History, 17.07.2019 14:50

History, 17.07.2019 14:50