
Chemistry, 18.03.2021 03:20 LarryJoeseph
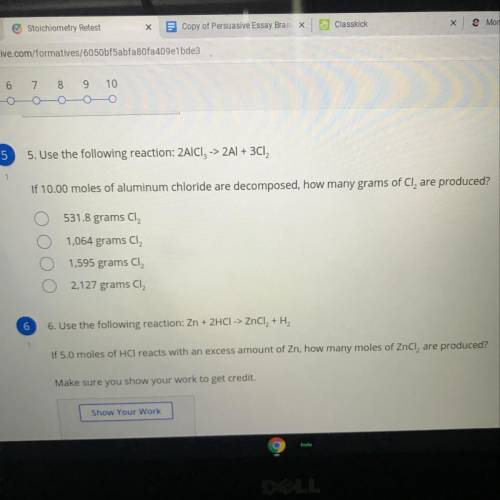
Use the following reaction: 2AICI, -> 2Al + 3Cl2
If 10.00 moles of aluminum chloride are decomposed, how many grams of Cl, are produced?
A. 531.8 grams Cl2
B. 1,064 grams CI2
C. 1,595 grams Cl2
D. 2,127 grams CI2

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
The earth's moon is unusually large. two popular theories of the moon's origin include the "sister world" hypothesis, which states that the moon formed from the same materials as the earth, near enough to the earth that they fell into orbit around each other. a second theory is the "capture" hypothesis, in which the moon formed elsewhere in the solar system, and the earth's gravity pulled it into its orbit. studies of what the moon is made of indicate that some of its materials had to come from the earth or from the same area of the solar system where the earth had formed. at the same time, the moon does not contain much of the material that makes up the earth's core, so the moon could not have formed from the same materials as the earth. how do the two facts above affect the described theories of the moon's origin? a. they show that scientists will never agree on where the moon came from. b. they show that more experiments on moon formation need to be done. c. they show that no theory accounts for the existence of the moon. d. they show that neither theory is complete and entirely correct.
Answers: 1

Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3


Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
Use the following reaction: 2AICI, -> 2Al + 3Cl2
If 10.00 moles of aluminum chloride are decompo...
Questions

Health, 06.07.2019 19:00

History, 06.07.2019 19:00





History, 06.07.2019 19:00


Mathematics, 06.07.2019 19:00


Mathematics, 06.07.2019 19:00

History, 06.07.2019 19:00

Chemistry, 06.07.2019 19:00

Mathematics, 06.07.2019 19:00



Biology, 06.07.2019 19:00

Mathematics, 06.07.2019 19:00

Mathematics, 06.07.2019 19:00



