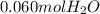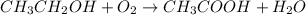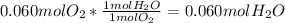Chemistry, 13.03.2021 04:50 keigleyhannah30
Wine goes bad soon after opening because Ethanol (CH3CH2OH) dissolved in it reacts with oxygen (O2) gas to form water and Aqueous Acetic acid (CH3COOH) The main ingredient of vinegar. Calculate the moles of water produced by the reaction of 0.060 mol of oxygen

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2


Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
Wine goes bad soon after opening because Ethanol (CH3CH2OH) dissolved in it reacts with oxygen (O2)...
Questions


Business, 08.10.2019 20:00









Mathematics, 08.10.2019 20:00



Chemistry, 08.10.2019 20:00

History, 08.10.2019 20:00

Mathematics, 08.10.2019 20:00


History, 08.10.2019 20:00

Chemistry, 08.10.2019 20:00

Mathematics, 08.10.2019 20:00