
Chemistry, 31.01.2020 05:46 lwilliams28
Which statement describes spontaneous processes?
proceed without continuous external influence and have an overall negative free energy change
proceed without continuous external influence and have an overall positive free energy change
proceed with continuous external influence and have an overall negative free energy change
proceed with continuous external influence and have an overall positive free energy change

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3


Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1
You know the right answer?
Which statement describes spontaneous processes?
proceed without continuous external influenc...
proceed without continuous external influenc...
Questions

Mathematics, 28.10.2020 16:40





Social Studies, 28.10.2020 16:40



Business, 28.10.2020 16:40




English, 28.10.2020 16:40




Mathematics, 28.10.2020 16:40



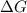 is negative.
is negative.

