
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxide gas, NO(g). This step is represented by the following balanced equation: 4 NH3(g) + 5 O2(g) --> 4 NO(g) + 6 H2O(l) Determine the amount of oxygen needed to produce 1.2 x 104 mol of nitrogen monoxide gas.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxi...
Questions



Social Studies, 19.08.2020 09:01



Mathematics, 19.08.2020 09:01





Mathematics, 19.08.2020 09:01



History, 19.08.2020 09:01

History, 19.08.2020 09:01

Mathematics, 19.08.2020 09:01


History, 19.08.2020 09:01


Physics, 19.08.2020 09:01

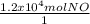 ×
× 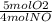 = 1.5 moles of O2 needed
= 1.5 moles of O2 needed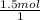 ×
× 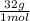 = 48 grams needed
= 48 grams needed

