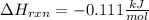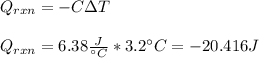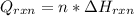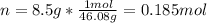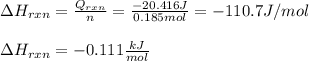Chemistry, 05.03.2021 23:00 sunpelt9993
An 8.5 g. Of ethanol (C2H5OH) placed in a constant volume calorimeter and the temperature rose to 3.2°C. Find the heat of reaction of the ethanol (MM =46.1 g/mol) in kJ/mol. The heat capacity of the calorimeter plus water is read as 6.38 J/°C

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1



You know the right answer?
An 8.5 g. Of ethanol (C2H5OH) placed in a constant volume calorimeter and the temperature rose to 3....
Questions



History, 19.03.2021 02:50






Biology, 19.03.2021 02:50


Mathematics, 19.03.2021 02:50

Mathematics, 19.03.2021 02:50

Mathematics, 19.03.2021 02:50