Chemistry, 04.03.2021 20:00 jennaranelli05
Calcium carbonate decomposes to form calcium oxide and carbon dioxide, like this:
CaCO3(s)→CaO(s)+CO2(g)
At a certain temperature, a chemist finds that a 9.0L reaction vessel containing a mixture of calcium carbonate, calcium oxide, and carbon dioxide at equilibrium has the following composition:
Compound Amount
CaCO3 25.3 g
CaO 14.9 g
CO2 33.7 g
Calculate the value of the equilibrium constant Kc for this reaction. Round your answer to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2

Chemistry, 22.06.2019 17:20
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2
You know the right answer?
Calcium carbonate decomposes to form calcium oxide and carbon dioxide, like this:
CaCO3(s)→CaO(s)+C...
Questions



History, 04.02.2020 13:58

Mathematics, 04.02.2020 13:58

English, 04.02.2020 13:58

Mathematics, 04.02.2020 13:58





Mathematics, 04.02.2020 13:58

English, 04.02.2020 13:59


Social Studies, 04.02.2020 13:59


Social Studies, 04.02.2020 13:59

Spanish, 04.02.2020 13:59


Mathematics, 04.02.2020 13:59

History, 04.02.2020 13:59

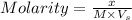
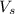 = volume of solution in L
= volume of solution in L
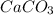 =
= 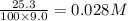
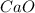 =
= 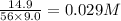
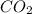 =
= 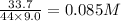
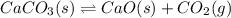
![K_c=\frac{[CaO]\times [CO_2]}{[CaCO_3]}](/tpl/images/1168/6891/ed17d.png)