
A buffer solution contains 0.345 M acetic acid and 0.377 M sodium acetate . If 0.0613 moles of potassium hydroxide are added to 250 mL of this buffer, what is the pH of the resulting solution ? (Assume that the volume does not change upon adding potassium hydroxide. )

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2


Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
A buffer solution contains 0.345 M acetic acid and 0.377 M sodium acetate . If 0.0613 moles of potas...
Questions


Mathematics, 11.11.2020 02:40

Biology, 11.11.2020 02:40


Mathematics, 11.11.2020 02:40




Geography, 11.11.2020 02:40



History, 11.11.2020 02:40

Mathematics, 11.11.2020 02:40



Mathematics, 11.11.2020 02:40



Social Studies, 11.11.2020 02:40

Mathematics, 11.11.2020 02:40

![\frac{[CH_3COO^-]}{[CH_3COOH]}](/tpl/images/1163/7311/4fc64.png)
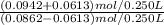 = 5.54
= 5.54

