3 points
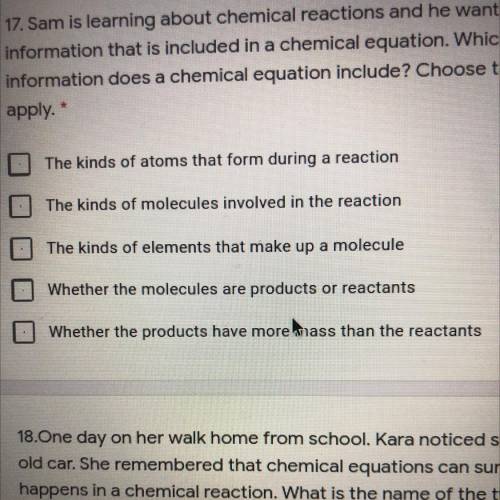
17. Sam is learning about chemical reactions and he wants to examine the
information...


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 23.06.2019 01:40
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1
You know the right answer?
Questions

Geography, 10.09.2020 02:01

Social Studies, 10.09.2020 02:01

Social Studies, 10.09.2020 02:01

Social Studies, 10.09.2020 02:01


Social Studies, 10.09.2020 02:01

Social Studies, 10.09.2020 02:01

English, 10.09.2020 02:01



Mathematics, 10.09.2020 02:01



History, 10.09.2020 02:01


Social Studies, 10.09.2020 02:01

Social Studies, 10.09.2020 02:01

Computers and Technology, 10.09.2020 02:01

Mathematics, 10.09.2020 02:01

History, 10.09.2020 02:01