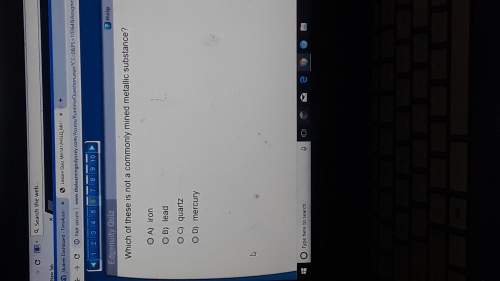
Chemistry, 04.01.2021 17:20 Kelseyyleigh2611
In one of his experiments, Lavoisier placed 10.0 grams of mercury (II) oxide into a sealed container and heated it. The mercury (II) oxide then reacted in the presence of heat to produce 9.3 grams of mercury. Oxygen gas was another product in the reaction. According to the law of conservation of mass, how much oxygen gas would have been produced?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
In one of his experiments, Lavoisier placed 10.0 grams of mercury (II) oxide into a sealed container...
Questions



Mathematics, 31.07.2019 05:00








Geography, 31.07.2019 05:10




Geography, 31.07.2019 05:10