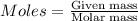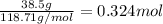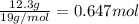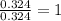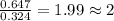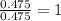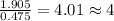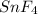Chemistry, 05.10.2019 16:20 devinluck100
Tin reacts with fluorine to form two different compounds, a and b. compound a contains 38.5 g of tin for each 12.3 g of fluorine. compound b contains 56.5 g of tin for each 36.2 g of fluorine. what is the lowest whole-number mass ratio of tin that combines with a given mass of fluorine?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 19:20
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1
You know the right answer?
Tin reacts with fluorine to form two different compounds, a and b. compound a contains 38.5 g of tin...
Questions



Advanced Placement (AP), 30.05.2020 22:04





Mathematics, 30.05.2020 22:04

Biology, 30.05.2020 22:04



Mathematics, 30.05.2020 22:04





Physics, 30.05.2020 22:04