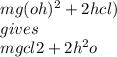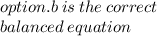Chemistry, 17.12.2020 20:40 nessuhbae6731
What is the complete balanced equation for the reaction
between magnesium hydroxide and hydrogen chloride to
produce magnesium chloride and water is
O MgOH + HCI → MgCl + H2O.
O Mg(OH)2 + 2HCl → MgCl2 + 2H20.
O Mg(OH)2 + HCl → Mg + 2CO2 +3H20.
O Mg(OH)2 + 2HCl → MgCl2 + H2 + O2.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 21:50
If e is the symbol for an element, which two of the following symbols represent isotopes of the same element? 1. e2. e3. ea.1 and 2c.1 and 4b.3 and 4d.2 and 3
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
What is the complete balanced equation for the reaction
between magnesium hydroxide and hydrogen ch...
Questions

History, 30.01.2021 21:20

Mathematics, 30.01.2021 21:20

Mathematics, 30.01.2021 21:20

Biology, 30.01.2021 21:20

Mathematics, 30.01.2021 21:20



Biology, 30.01.2021 21:20


Mathematics, 30.01.2021 21:20

Mathematics, 30.01.2021 21:20


Mathematics, 30.01.2021 21:20

Mathematics, 30.01.2021 21:20


Mathematics, 30.01.2021 21:20


History, 30.01.2021 21:20