
Chemistry, 17.12.2020 20:40 tshort2000oyreb1
I NEED HELP :(( Will mark brainliest i need the right answer these finals killing me... especially chem
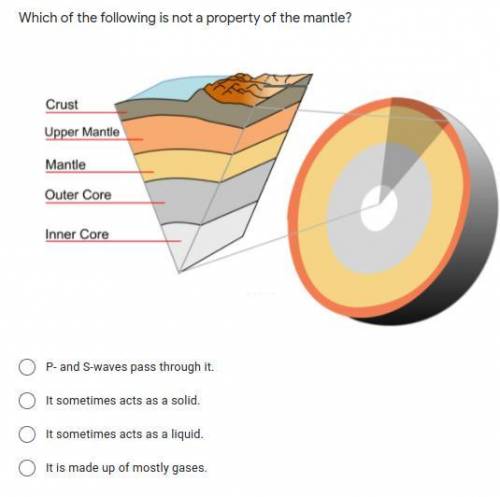

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

You know the right answer?
I NEED HELP :(( Will mark brainliest i need the right answer these finals killing me... especially...
Questions



Social Studies, 21.11.2020 14:00


Mathematics, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00


Biology, 21.11.2020 14:00

Law, 21.11.2020 14:00

Chemistry, 21.11.2020 14:00



Mathematics, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00




Health, 21.11.2020 14:00

History, 21.11.2020 14:00




