
Chemistry, 03.12.2020 14:20 babyduckies37
25.0cm3 of s saturated potassium hydroxide is neutralized by 35.0cm3 of hydrogen chloride acid of concentration 0.75 mol/dm3. Calculate the concentration of potassium hydroxide solution. Please help, will give brainliest, unhelpful answers will get reported.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:40
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3
You know the right answer?
25.0cm3 of s saturated potassium hydroxide is neutralized by 35.0cm3 of hydrogen chloride acid of co...
Questions



Spanish, 08.11.2020 04:20


Mathematics, 08.11.2020 04:20

Social Studies, 08.11.2020 04:20


English, 08.11.2020 04:30

Mathematics, 08.11.2020 04:30


Physics, 08.11.2020 04:30


Health, 08.11.2020 04:30

Mathematics, 08.11.2020 04:30

Mathematics, 08.11.2020 04:30


Mathematics, 08.11.2020 04:30



English, 08.11.2020 04:30

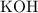 solution: approximately
solution: approximately 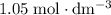 .
. 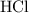 solution is in the unit
solution is in the unit 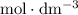 . However, the unit of the two volumes is
. However, the unit of the two volumes is 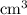 . Convert the unit of the two volumes to
. Convert the unit of the two volumes to 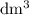 to match the unit of concentration.
to match the unit of concentration.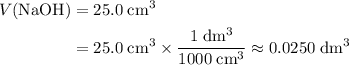 .
.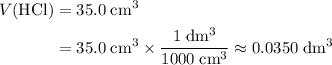 .
.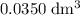 of
of 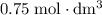
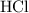 solution:
solution: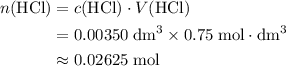 .
.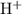 .
.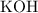 formula unit would react with up to one
formula unit would react with up to one 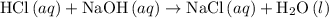 .
.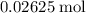 of
of 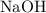 formula units. That is:
formula units. That is: 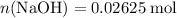 .
.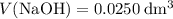 and
and 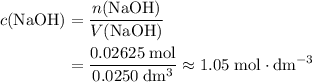 .
.

