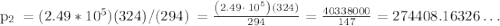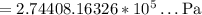Chemistry, 25.08.2020 01:01 alyssalefeber
If anyone is good at chemistry do you mind helping :)


Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
If anyone is good at chemistry do you mind helping :)
...
...
Questions


English, 27.08.2020 09:01




Mathematics, 27.08.2020 09:01

Physics, 27.08.2020 09:01


Mathematics, 27.08.2020 09:01


Mathematics, 27.08.2020 09:01

Chemistry, 27.08.2020 09:01

Arts, 27.08.2020 09:01


Chemistry, 27.08.2020 09:01

English, 27.08.2020 09:01

Mathematics, 27.08.2020 09:01



Computers and Technology, 27.08.2020 09:01

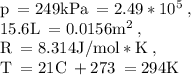
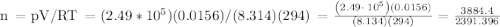
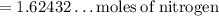
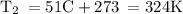 -
-