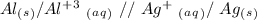Chemistry, 12.08.2020 08:01 jothianddeepi
Ag2S + Al(s) = Al2S3 + Ag(s) (unbalanced)
1) What would the overall potential for this cell be?
2) Write the standard cell notation for an electrochemical cell consisting of an anode and cathode of the same types as in this experiment, connected through a salt bridge.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3
You know the right answer?
Ag2S + Al(s) = Al2S3 + Ag(s) (unbalanced)
1) What would the overall potential for this cell be?
Questions



English, 30.11.2019 04:31



Biology, 30.11.2019 04:31

Biology, 30.11.2019 04:31


Biology, 30.11.2019 04:31

Biology, 30.11.2019 04:31



Chemistry, 30.11.2019 04:31

Biology, 30.11.2019 04:31






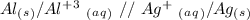
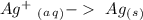
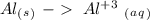
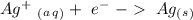 Reduction
Reduction 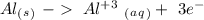 Oxidation
Oxidation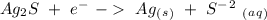 - 0.69 V
- 0.69 V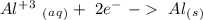 -1.66 V
-1.66 V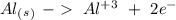 +1.66 V
+1.66 V