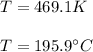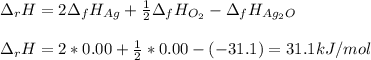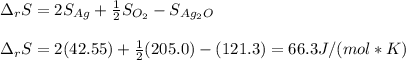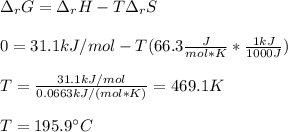Chemistry, 12.08.2020 05:01 jetjet123123
Given the data: Ag2O(s), = ‑31.1 kJ mol-1, S° = +121.3 J mol-1 K-1 Ag(s), = 0.00 kJ mol-1, S° = +42.55 J mol-1 K-1 O2(g), = 0.00 kJ mol-1, S° = +205.0 J mol-1 K-1 Calculate the temperature at which = 0 for the reaction, Ag2O(s) → 2 Ag(s) + ½ O2(g). Assume that, since the physical states do not change, and are independent of temperature between ‑50.0 °C and 950.0 °C.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2
You know the right answer?
Given the data: Ag2O(s), = ‑31.1 kJ mol-1, S° = +121.3 J mol-1 K-1 Ag(s), = 0.00 kJ mol-1, S° = +42....
Questions

Mathematics, 20.10.2019 18:50


Social Studies, 20.10.2019 18:50

English, 20.10.2019 18:50

Physics, 20.10.2019 18:50


Mathematics, 20.10.2019 18:50


History, 20.10.2019 18:50

Mathematics, 20.10.2019 18:50



Mathematics, 20.10.2019 18:50



Mathematics, 20.10.2019 18:50



Social Studies, 20.10.2019 18:50