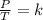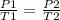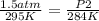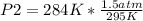Chemistry, 31.07.2020 23:01 saneayahsimmons
Given a fixed amount of gas in a rigid container (no change in volume), what pressure will the gas exert if the pressure is initially 1.50 atm at 22.0oC, and the temperature is changed to 11.0oC?
A. 301 atm
B. 1.56 atm
C. 0.750 atm
D. 1.44 atm
E. 3.00 atm

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 22.06.2019 03:00
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
Given a fixed amount of gas in a rigid container (no change in volume), what pressure will the gas e...
Questions

Biology, 13.10.2019 07:50

History, 13.10.2019 07:50

Mathematics, 13.10.2019 07:50




Physics, 13.10.2019 07:50

English, 13.10.2019 07:50

English, 13.10.2019 07:50

Mathematics, 13.10.2019 07:50


Mathematics, 13.10.2019 07:50


Physics, 13.10.2019 07:50


Mathematics, 13.10.2019 07:50

History, 13.10.2019 07:50

Chemistry, 13.10.2019 07:50


Mathematics, 13.10.2019 07:50