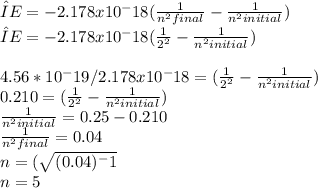Chemistry, 15.07.2020 01:01 cutekute22
A transition in the balmer series for hydrogen has an observed wavelength of 434 nm. Use the Rydberg equation below to find the energy level that the transition originated. Transitions in the Balmer series all terminate n=2.
Delta E= -2.178 x10-18J ( 1/n2Final - 1/n2Initial )
The number is 5.
What is the energy of this transition in units of kJ/mole? ( hint: the anser is NOT 4.58x10-22kJ/mole or -4.58x10-22kJ/mole)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1


Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1
You know the right answer?
A transition in the balmer series for hydrogen has an observed wavelength of 434 nm. Use the Rydberg...
Questions

Mathematics, 16.01.2020 10:31

Mathematics, 16.01.2020 10:31

Mathematics, 16.01.2020 10:31


Mathematics, 16.01.2020 10:31


Mathematics, 16.01.2020 10:31



Biology, 16.01.2020 10:31


Biology, 16.01.2020 10:31


Mathematics, 16.01.2020 10:31


Mathematics, 16.01.2020 10:31




Mathematics, 16.01.2020 10:31

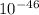 KJ/mole
KJ/mole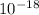 (
(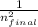 -
- 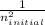 )
)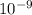 ) = -2.178 ×
) = -2.178 × 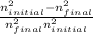 )
)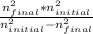
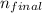 = 2
= 2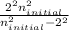
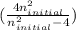
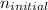 = 5
= 5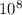 ) ÷ (434 ×
) ÷ (434 × 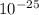 ) ÷ (434 ×
) ÷ (434 × 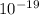 J
J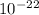 KJ
KJ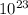 , then;
, then;