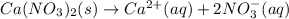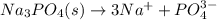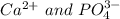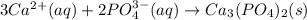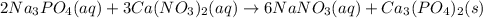Chemistry, 28.06.2020 07:01 woodsjnjoseph3
During lab, students mixed two solutions of soluble ions in a ceramic well to determine if a precipitate forms.
Write the dissolution reaction for the ionic solids below. (Use the lowest possible coefficients. Include states-of-matter under the given conditions in your answer.)
(a) Ca(NO3)2
(b) Na3PO4
The two solutions, when mixed, will have two cations and two anions.
(c) Based on your lab results, enter the cation and anion for which a precipitate will form. (Separate substances in a list with a comma.)
(d) Write the net precipitation reaction that occurs. (Use the lowest possible coefficients. Include states-of-matter under the given conditions in your answer.)

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1


Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
You know the right answer?
During lab, students mixed two solutions of soluble ions in a ceramic well to determine if a precipi...
Questions

German, 24.01.2021 23:40



Mathematics, 24.01.2021 23:40











Biology, 24.01.2021 23:40

Mathematics, 24.01.2021 23:40

Mathematics, 24.01.2021 23:40


Mathematics, 24.01.2021 23:40