Chemistry, 23.06.2020 10:57 mvtthewisdead
The reaction 2 NO(g) ⇌ N2(g) + O2(g) has a value of Keq = 2400 at a temperature of 2000 K. If 0.570 mol of NO(g) is initially placed in a 3.0 L container, calculate the equilibrium concentrations of each gas

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1


Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1

Chemistry, 23.06.2019 02:00
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2
You know the right answer?
The reaction 2 NO(g) ⇌ N2(g) + O2(g) has a value of Keq = 2400 at a temperature of 2000 K. If 0.570...
Questions









Mathematics, 06.09.2019 03:30




Mathematics, 06.09.2019 03:30



Computers and Technology, 06.09.2019 03:30




![[N_2]_{eq}=0.094M](/tpl/images/0692/1303/f5b19.png)
![[O_2]_{eq}=0.094M](/tpl/images/0692/1303/b20e2.png)
![[NO]_{eq}=0.002M](/tpl/images/0692/1303/096e8.png)
![Keq=\frac{[N_2][O_2]}{[NO]^2}](/tpl/images/0692/1303/93add.png)
 due to the reaction extent (ICE procedure) we can write:
due to the reaction extent (ICE procedure) we can write:![Keq=\frac{x*x}{([NO]_0-2x)^2}](/tpl/images/0692/1303/d6f53.png)
![[NO]_0=\frac{0.570mol}{3.0L} =0.19M](/tpl/images/0692/1303/2b637.png)
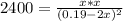
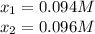
![[N_2]_{eq}=x=0.094M](/tpl/images/0692/1303/b3325.png)
![[O_2]_{eq}=x=0.094M](/tpl/images/0692/1303/3c9f2.png)
![[NO]_{eq}=0.19M-2x=0.19M-2(0.094M)=0.002M](/tpl/images/0692/1303/c2eca.png)