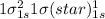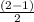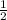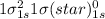Chemistry, 21.05.2020 05:10 korireidkdotdot82021
Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. The He2 + ion is more stable since it has a higher bond order (bond order = 1) than the He2 2+ ion (bond order = 1/2). The He2 + ion is more stable since it has a higher bond order (bond order = 2) than the He2 2+ ion (bond order = 1). The He2 2+ ion is more stable since it has a lower bond order (bond order = 1/2) than the He2 + ion (bond order = 1). The He2 2+ a lower bond order (bond order = 3/2) than the He2 + ion (bond order = 2). The He2 2+ ion is more stable since it has a higher bond order (bond order = 1) than the He2 + ion (bond order = 1/2).

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:30
Which is the most likely way an automotive engineer would use chemistry
Answers: 1

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
You know the right answer?
Use molecular orbital theory to determine whether He2 2+ or He2 + is more stable. Use molecular orbi...
Questions

History, 22.02.2021 19:30

Mathematics, 22.02.2021 19:30



Law, 22.02.2021 19:30



Chemistry, 22.02.2021 19:30

English, 22.02.2021 19:30




Mathematics, 22.02.2021 19:30

Mathematics, 22.02.2021 19:30

Social Studies, 22.02.2021 19:30


Biology, 22.02.2021 19:30



Mathematics, 22.02.2021 19:30