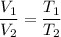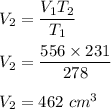Chemistry, 21.05.2020 03:00 lexiscaraballo12
A student decreases the temperature of a 556 c m cubed balloon from 278 K to 231 K. Assuming constant pressure, what should the new volume of the balloon be?
Question 1 options:
924cm3
417cm3
347cm3
876cm3

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:30
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3

Chemistry, 22.06.2019 00:30
Lem 2 the data below are for the system ethyl propyl ether (1)-chloroform (2) at 0.5 bar. use the data to answer the following questions (all questions refer to p d 0: 5 bar). a) what are the boiling points of the pure components at 0.5 bar? b) a mixture with the overall composition z1 d 0: 1 is brought to 47.6ä±c, 0.5 bar. what is the phase? c) 100 mole of a mixture with z1 d 0: 1 (state a) is mixed with 22 mole of pure ethyl propyl ether vapor (state b). the mixing takes place at 47.6 ä±c, 0.5. bar. what is the phase of the resulting mixture (state c)? if the state is a v/l mixture report the number of moles and mole fractions in each phase. d) plot the txy graph and show states a, b and c. the graph must be done by computer and should be properly annotated. ethyl propyl ether (1) - chloroform (2) at 0.5 bar t ( ä±c) x1 y1 t ( ä±c) x1 y1 42.9 0.000 0.000 49.0 0.470 0.455 43.0 0.020 0.010 49.1 0.520 0.520 43.9 0.065 0.029 48.9 0.567 0.592 45.4 0.156 0.089 48.3 0.652 0.720 46.4 0.215 0.142 47.6 0.745 0.815 47.6 0.296 0.223 46.7 0.822 0.872 48.3 0.362 0.302 45.7 0.907 0.937 48.7 0.410 0.375 44.6 1.000
Answers: 3

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1
You know the right answer?
A student decreases the temperature of a 556 c m cubed balloon from 278 K to 231 K. Assuming constan...
Questions


Mathematics, 22.08.2019 02:00

Mathematics, 22.08.2019 02:00

Physics, 22.08.2019 02:00

Mathematics, 22.08.2019 02:00


Business, 22.08.2019 02:00



Social Studies, 22.08.2019 02:00

History, 22.08.2019 02:00


Mathematics, 22.08.2019 02:00


History, 22.08.2019 02:00

Biology, 22.08.2019 02:00


Social Studies, 22.08.2019 02:00

Mathematics, 22.08.2019 02:00

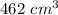 .
. 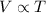 , P is constant
, P is constant