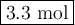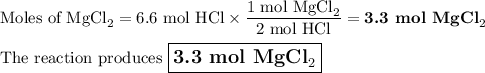Chemistry, 22.04.2020 05:28 Camill0310
If 6.6 moles of HCI reacts with excess magnesium, how many moles of magnesium chloride will be produced?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
If 6.6 moles of HCI reacts with excess magnesium, how many moles of magnesium chloride will be produ...
Questions

Health, 17.01.2020 01:31


English, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Computers and Technology, 17.01.2020 01:31


Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31


Mathematics, 17.01.2020 01:31


Mathematics, 17.01.2020 01:31


Advanced Placement (AP), 17.01.2020 01:31

English, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31