Chemistry, 15.04.2020 00:52 calebcoolbeans6691
A chemist titrates 160.0 mL of a 0.3065 M cyanic acid (HCNO) solution with 0.4994 M NaOH solution at 25 °C. Calculate the pH at equivalence. The pK of cyanic acid is 3.46 Round your answer to 2 decimal places.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 23:30
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3

Chemistry, 23.06.2019 01:50
Drag the tiles to the correct locations. each tile can be used more than once, but not all tiles will be used. one or more locations will remain empty. nitrosyl fluoride has the chemical formula nof nitrogen has five valence electrons, oxygen has six, and fluorine has seven. complete the lewis structure for this covalent compound. f n = = = . : : 0 : reset next um. all rights reserved us 2
Answers: 2

Chemistry, 23.06.2019 06:00
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2
You know the right answer?
A chemist titrates 160.0 mL of a 0.3065 M cyanic acid (HCNO) solution with 0.4994 M NaOH solution at...
Questions

Biology, 13.07.2019 17:00


Geography, 13.07.2019 17:00


Biology, 13.07.2019 17:00

Biology, 13.07.2019 17:00

Computers and Technology, 13.07.2019 17:00

History, 13.07.2019 17:00





Health, 13.07.2019 17:00




English, 13.07.2019 17:00

Physics, 13.07.2019 17:00

Geography, 13.07.2019 17:00

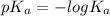
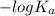
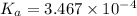
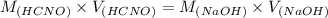
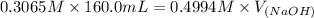
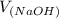 = 98.1978 mL
= 98.1978 mL
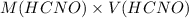
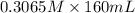
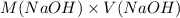
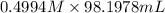
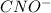 and
and 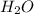 .
.
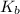 of
of 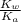
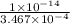

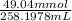
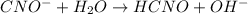
![K_{b} = \frac{[HCNO][OH^{-}]}{[CNO^{-}]}](/tpl/images/0600/3810/7fa50.png)
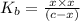
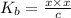
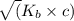
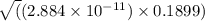
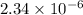
![[OH^{-}] = x = 2.34 \times 10^{-6}](/tpl/images/0600/3810/bb369.png) M
M
![-log [OH^{-}]](/tpl/images/0600/3810/337a4.png)