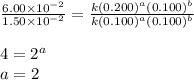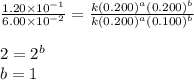Chemistry, 15.04.2020 01:53 beautycutieforever10
For the reaction 3A(g) + 2B(g) → 2C(g) + 2D(g) the following data were collected at constant temperature. Determine the correct rate law for this reaction. Trial Initial [A] Initial [B] Initial Rate (mol/L) (mol/L) (mol/(L·min)) 1 0.200 0.100 6.00 × 10–2 2 0.100 0.100 1.50 × 10–2 3 0.200 0.200 1.20 × 10–1 4 0.300 0.200 2.70 × 10–1

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2

Chemistry, 23.06.2019 09:30
What is the force of an object when it landed(sitting in the ground)
Answers: 2
You know the right answer?
For the reaction 3A(g) + 2B(g) → 2C(g) + 2D(g) the following data were collected at constant tempera...
Questions



Social Studies, 10.12.2020 01:10

Mathematics, 10.12.2020 01:10

Mathematics, 10.12.2020 01:10









Mathematics, 10.12.2020 01:10



English, 10.12.2020 01:10


English, 10.12.2020 01:10

![\text{Rate}=k[A]^2[B]^1](/tpl/images/0600/6826/858f4.png)
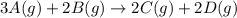
![\text{Rate}=k[A]^a[B]^b](/tpl/images/0600/6826/10aeb.png)
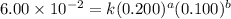 ....(1)
....(1)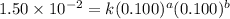 ....(2)
....(2)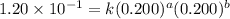 ....(3)
....(3)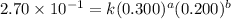 ....(4)
....(4)