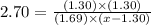For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) SO2(g)+NO2(g)↽−−⇀SO3(g)+NO(g) the equilibrium constant at a certain temperature is 2.70 . 2.70. At this temperature, calculate the number of moles of NO 2 ( g ) NO2(g) that must be added to 2.99 mol SO 2 ( g ) 2.99 mol SO2(g) in order to form 1.30 mol SO 3 ( g ) 1.30 mol SO3(g) at equilibrium.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1


Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) SO2(g)+NO2(g)↽−−⇀SO3...
Questions





Mathematics, 18.01.2021 21:50


Business, 18.01.2021 21:50













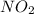 added must be, 0.37 mol
added must be, 0.37 mol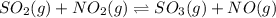
![K_c=\frac{[SO_3][NO]}{[SO_2][NO_2]}](/tpl/images/0599/9503/d13ef.png)