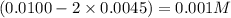Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
How many ions that have a +1 charge will bond with an ion that has a -2 charge
Answers: 1

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1
You know the right answer?
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however,...
Questions



Mathematics, 30.12.2021 03:30

English, 30.12.2021 03:30


Physics, 30.12.2021 03:30


Computers and Technology, 30.12.2021 03:30




Social Studies, 30.12.2021 03:30

Mathematics, 30.12.2021 03:30

Chemistry, 30.12.2021 03:30



Mathematics, 30.12.2021 03:30

Business, 30.12.2021 03:30

Chemistry, 30.12.2021 03:30

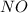 at equilibrium = 0.001 M
at equilibrium = 0.001 M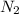 = 0.0045 M
= 0.0045 M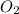 = 0.0045 M
= 0.0045 M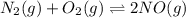
![K_c=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0599/1341/71f8f.png)
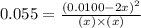
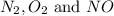 at equilibrium are :
at equilibrium are :