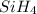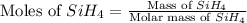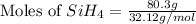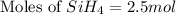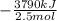Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Write the symbol for every chemical element that has atomic number greater than 3 and atomic mass less than 12.0 u.
Answers: 1

Chemistry, 21.06.2019 22:30
Using the periodic table, complete the table to describe each atom. type in your answers.a ? b? c? d? e? f?
Answers: 1


Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
The burning of 80.3 g of SiH4 at constant pressure gives off 3790 kJ of heat. Calculate △H for this...
Questions


Computers and Technology, 02.10.2019 22:30




English, 02.10.2019 22:30

Mathematics, 02.10.2019 22:30




Health, 02.10.2019 22:30



Mathematics, 02.10.2019 22:30



History, 02.10.2019 22:30



Biology, 02.10.2019 22:30