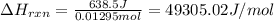Chemistry, 31.01.2020 02:45 rileyeddins1010
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride according to the reaction: agno3(aq)+hcl(aq)→agcl(s)+hno3(aq). when you combine 70.0ml of 0.185m agno3 with 70.0ml of 0.185m hcl in a coffee-cup calorimeter, the temperature changes from 23.16∘c to 24.25∘c.. calculate δhrxn for the reaction as written. use 1.00 g/ml as the density of the solution and c=4.18j/g⋅∘c as the specific heat capacity.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride accordin...
Questions













Computers and Technology, 10.12.2019 05:31







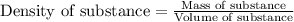
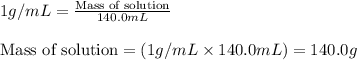
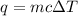
 = change in temperature =
= change in temperature = ![[24.25-23.16]^oC=1.09^oC](/tpl/images/0488/1311/2253a.png)
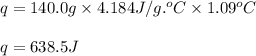
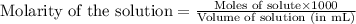
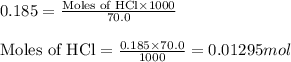
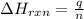
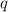 = amount of heat absorbed = 638.5 J
= amount of heat absorbed = 638.5 J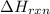 = enthalpy change of the reaction
= enthalpy change of the reaction