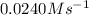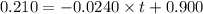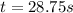Chemistry, 07.04.2020 15:59 angelricardoblp8kwg3
The rate constant for this zero‑order reaction is 0.0240 M ⋅ s − 1 0.0240 M·s−1 at 300 ∘ C. 300 ∘C. A ⟶ products A⟶products How long (in seconds) would it take for the concentration of A A to decrease from 0.900 M 0.900 M to 0.210 M?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Identify which properties could correspond to solids, plasmas, or both. maintain a unique shape. collide infrequently with other particles. have very high velocities. conduct electricity. protons. have a low temperature. has long-range order.
Answers: 1



Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
The rate constant for this zero‑order reaction is 0.0240 M ⋅ s − 1 0.0240 M·s−1 at 300 ∘ C. 300 ∘C....
Questions

Social Studies, 30.11.2019 20:31

History, 30.11.2019 20:31


History, 30.11.2019 20:31




History, 30.11.2019 20:31

Physics, 30.11.2019 20:31





History, 30.11.2019 20:31




Physics, 30.11.2019 20:31


![[A]=-kt+[A]_o](/tpl/images/0586/4094/d191d.png)
![[A]_o](/tpl/images/0586/4094/9caf5.png) = initial concentration = 0.900 M
= initial concentration = 0.900 M