Chemistry, 04.04.2020 10:39 lucky1silky
A 30.5-g sample of an alloy at 93.0°C is placed into 50.0 g of water at 22.0°C in an insulated coffee-cup calorimeter with a heat capacity of 9.2 J/K. If the final temperature of the system is 31.1°C, what is the specific heat capacity of the alloy?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 22.06.2019 23:00
Which organism develops breathing organism develops breathing organs from pharyngeal arches? shark, spider, sea star, sea horse
Answers: 2
You know the right answer?
A 30.5-g sample of an alloy at 93.0°C is placed into 50.0 g of water at 22.0°C in an insulated coffe...
Questions

Mathematics, 16.10.2020 18:01

History, 16.10.2020 18:01

Mathematics, 16.10.2020 18:01




Geography, 16.10.2020 18:01

Mathematics, 16.10.2020 18:01

Chemistry, 16.10.2020 18:01

Physics, 16.10.2020 18:01

Biology, 16.10.2020 18:01


Mathematics, 16.10.2020 18:01


History, 16.10.2020 18:01


Biology, 16.10.2020 18:01

English, 16.10.2020 18:01


Social Studies, 16.10.2020 18:01

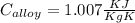
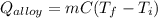
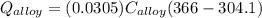
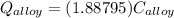 ------- (1)
------- (1)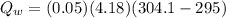
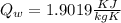 ------- (2)
------- (2)