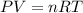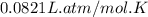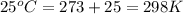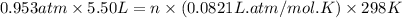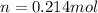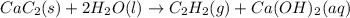Chemistry, 31.03.2020 00:57 MajentaSnow66
Calcium carbide reacts with water to produce acetylene gas according to the following equation: CaC2(s) + 2H2O(l)C2H2(g) + Ca(OH)2(aq) The product gas, C2H2, is collected over water at a temperature of 25 °C and a pressure of 748 mm Hg. If the wet C2H2 gas formed occupies a volume of 5.50 L, the number of moles of CaC2 reacted was mol. The vapor pressure of water is 23.8 mm Hg at 25 °C.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
Calcium carbide reacts with water to produce acetylene gas according to the following equation: CaC2...
Questions














Mathematics, 17.03.2020 02:20




Biology, 17.03.2020 02:20


Mathematics, 17.03.2020 02:20

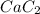 reacted was, 0.214 moles.
reacted was, 0.214 moles.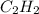 gas.
gas.